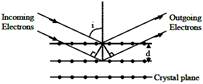
 Electrons accelerated by potential V are diffracted from a crystal. If \[d=1\overset{o}{\mathop{A}}\,\] and\[i={{30}^{o}}\], V should be about (\[h=6.6\times {{10}^{-34}}Js\],\[{{m}_{e}}=9.1\times {{10}^{-31}}kg,\,e=1.6\times {{10}^{-19}}C\])
AIEEE Solved Paper-2007
Electrons accelerated by potential V are diffracted from a crystal. If \[d=1\overset{o}{\mathop{A}}\,\] and\[i={{30}^{o}}\], V should be about (\[h=6.6\times {{10}^{-34}}Js\],\[{{m}_{e}}=9.1\times {{10}^{-31}}kg,\,e=1.6\times {{10}^{-19}}C\])
AIEEE Solved Paper-2007
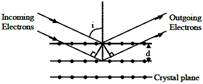 If a strong diffraction peak is observed when electrons are incident at an angle 'I' from the normal to the crystal planes with distance 'd' between them (see figure), de Broglie wavelength \[{{\lambda }_{dB}}\] of electrons can be calculated by the relationship (n is an integer)
AIEEE Solved Paper-2007
If a strong diffraction peak is observed when electrons are incident at an angle 'I' from the normal to the crystal planes with distance 'd' between them (see figure), de Broglie wavelength \[{{\lambda }_{dB}}\] of electrons can be calculated by the relationship (n is an integer)
AIEEE Solved Paper-2007
You need to login to perform this action.
You will be redirected in
3 sec
