A) Ne
B) Ar
C) Xe
D) Kr
Correct Answer: C
Solution :
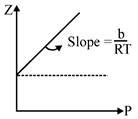 \[As\,b\uparrow \Rightarrow slope\,\uparrow \] Hence, Xe, will have highest slope
\[As\,b\uparrow \Rightarrow slope\,\uparrow \] Hence, Xe, will have highest slope
You need to login to perform this action.
You will be redirected in
3 sec
