A) 2-Hydroxycyclopentanone
B) 1-Pentanol
C) 3-Hydroxypentan-2-one
D) 4-Hydroxypentan-2-one
Correct Answer: C , D
Solution :
3-Hydroxypentan-2-one and 4-Hydroxypentan-2-one will most readily be dehydrated to give alkene under acidic condition. This will lead to formation of \[\alpha -\beta \]unsaturated ketone. Note: Dehydration of 2-Hydroxycyclopentanone will lead to C = C double bond in 5 member ring that will cause strain and product is less likely to be formed.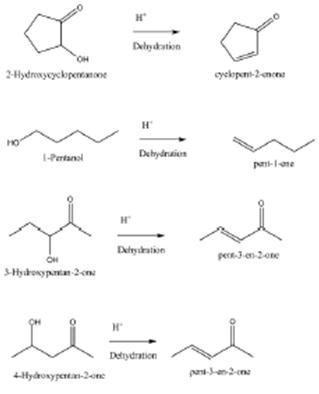
Solution :
3-Hydroxypentan-2-one and 4-Hydroxypentan-2-one will most readily be dehydrated to give alkene under acidic condition. This will lead to formation of \[\alpha -\beta \]unsaturated ketone. Note: Dehydration of 2-Hydroxycyclopentanone will lead to C = C double bond in 5 member ring that will cause strain and product is less likely to be formed.
You need to login to perform this action.
You will be redirected in
3 sec
