A) 2.2 ms
B) 5 ms
C) 3.3 ms
D) 7.2 ms
Correct Answer: C
Solution :
Suppose 'm' gram of water evaporates then, heat required \[\Delta {{Q}_{req}}=m{{L}_{\text{v}}}\] Mass that converts into ice = (150 - m) So, heat released in this process \[\Delta {{Q}_{rel}}=(150-m){{L}_{f}}\] Now, \[\Delta {{Q}_{rel}}=\Delta {{Q}_{req}}\] \[(150-m){{L}_{f}}=m{{L}_{V}}\] \[m({{L}_{f}}+{{L}_{\text{v}}})=150{{L}_{f}}\] \[m=\frac{150{{L}_{f}}}{{{L}_{f}}+{{L}_{\text{v}}}}\] \[m=20g\]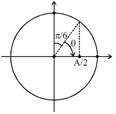 \[V\left( t \right)=220\text{ }sin(100\pi t)\text{ }volt\] time taken, \[t=\frac{\theta }{\omega }=\frac{\frac{\pi }{3}}{100\pi }=\frac{1}{300}\sec \]\[=3.3ms\]
\[V\left( t \right)=220\text{ }sin(100\pi t)\text{ }volt\] time taken, \[t=\frac{\theta }{\omega }=\frac{\frac{\pi }{3}}{100\pi }=\frac{1}{300}\sec \]\[=3.3ms\]
You need to login to perform this action.
You will be redirected in
3 sec
