A) octahedral
B) pentagonal planar
C) square pyramidal
D) trigonal bipyramidal
Correct Answer: C
Solution :
In \[XeO{{F}_{4}},Xe\]is \[s{{p}^{3}}{{d}^{2}},\] hybridised having 5 Bond pairs & 1 lone pair SHAPE: Square Pyramidal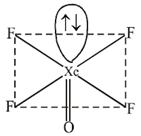
You need to login to perform this action.
You will be redirected in
3 sec
