A) \[{{(Si{{H}_{3}})}_{3}}N\]is pyramidal and more basic than \[{{(C{{H}_{3}})}_{3}}N\]
B) \[{{(Si{{H}_{3}})}_{3}}N\] is planar and more basic than\[{{(C{{H}_{3}})}_{3}}N\]
C) \[{{(Si{{H}_{3}})}_{3}}N\]is pyramidal and less basic than\[{{(C{{H}_{3}})}_{3}}N\]
D) \[{{(Si{{H}_{3}})}_{3}}N\] is planar and less basic than\[{{(C{{H}_{3}})}_{3}}N\]
Correct Answer: D
Solution :
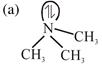 nitrogen is \[s{{p}^{3}}\]hybrid and pyramidal no back-bonding i.e. more basic
nitrogen is \[s{{p}^{3}}\]hybrid and pyramidal no back-bonding i.e. more basic  Nitrogen \[s{{p}^{2}}\]hybrid and planar due to back bonding and less basic because lone pair is not available for donation.
Nitrogen \[s{{p}^{2}}\]hybrid and planar due to back bonding and less basic because lone pair is not available for donation.
You need to login to perform this action.
You will be redirected in
3 sec
