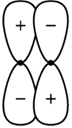
A) A bonding \[\pi \] orbital
B) A non-bonding orbital
C) An antibonding \[\sigma \] orbital
D) An antibonding \[\pi \] orbital
Correct Answer: D
Solution :
An antibonding \[\pi \] orbital best describes the diagram of a molecular orbital. Two p orbitals laterally overlap to form pi bond. Out of phase combination of these two p orbitals give \[\text{ }\!\!\pi\!\!\text{ *MO}\].You need to login to perform this action.
You will be redirected in
3 sec
