A) \[s{{p}^{3}},\] two
B) \[ds{{p}^{2}},\]zero
C) \[ds{{p}^{2}},\]one
D) \[s{{p}^{3}},\]zero
Correct Answer: A
Solution :
\[{{[Ni{{L}_{4}}]}^{2-}}\]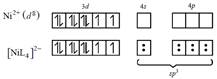 i.e, no. of unpaired electron = 2 hybridization \[-s{{p}^{3}}.\]
i.e, no. of unpaired electron = 2 hybridization \[-s{{p}^{3}}.\]
You need to login to perform this action.
You will be redirected in
3 sec
