| (i) It arises due to vander Waals forces and |
| (ii) it is reversible. Identify the correct statement that describes the above adsorption process: |
A) Enthalpy of adsorption is greater than \[100 \text{kJ}\,\text{mo}{{\text{l}}^{\text{-1}}}.\]
B) Adsorption is monolayer.
C) Adsorption increases with increase in temperature.
D) Energy of activation is low.
Correct Answer: D
Solution :
Adsorption aries due to Vander waal forces & reversible, hence it should be physisorption (physical adsorption). (i) Enthalpy of physisorption is low (20 . 40 kJ/mol) (ii)In physisorption multimolecular layer form. (iii)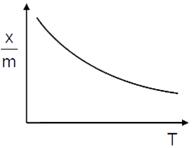 Physisorption decreases with increase in temperature. (iv) Physisorption required number activation energy. Hence answer is (d)
Physisorption decreases with increase in temperature. (iv) Physisorption required number activation energy. Hence answer is (d)
You need to login to perform this action.
You will be redirected in
3 sec
