| A gas can be taken from A to B via two different processes ACB and ADB. |
| [JEE Main 09-Jan-2019 Morning] |
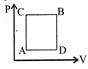 |
| When path ACB is used 60 J of heat flows into the system and 30 J of work is done by the system. If path ADB is used work done by the system is 10 J. The heat Flow into the system in path ADB is: |
A) 40 J
B) 20 J
C) 100 J
D) 80 J
Correct Answer: A
Solution :
| [a] |
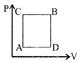 |
| For Path ACB |
| \[Q\text{ }=\text{ }60\text{ }J\] |
| \[W\text{ }=\text{ }30\text{ }J\] |
| \[\because \,\,\,\,Q\text{ }=\text{ }U\text{ }+\text{ }W\] |
| \[\Rightarrow \,\,\,U\text{ }=\text{ }30\text{ }J\] |
| Now, \[{{U}_{ACB}}\,\,=\,\,{{U}_{ADB}}\,\,=\,\,30\text{ }J\] |
| For Path ADB:- |
| \[U\,\,\,=\,\,30\text{ }J\] |
| \[W=10\,J\] |
| \[\Rightarrow \,\,\,Q=U+W=40\,J\] |
| Option [a] is correct. |
You need to login to perform this action.
You will be redirected in
3 sec
