A) trigonal bipyramidal
B) tetrahedral
C) octahedral
D) square planar
Correct Answer: D
Solution :
\[{}_{29}Cu=1{{s}^{2}},2{{s}^{2}}2{{p}^{6}},3{{s}^{2}}3{{p}^{6}}3{{d}^{10}},4{{s}^{1}}\] \[C{{u}^{2+}}=[Ar]\,3{{d}^{9}},4{{s}^{0}}4{{p}^{0}}\]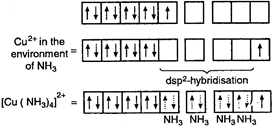 Hence, geometry of \[{{[Cu{{(N{{H}_{3}})}_{4}}]}^{2+}}\] ion is square-planar and it is paramagnetic ion, as it has one unpaired electron.
Hence, geometry of \[{{[Cu{{(N{{H}_{3}})}_{4}}]}^{2+}}\] ion is square-planar and it is paramagnetic ion, as it has one unpaired electron.
You need to login to perform this action.
You will be redirected in
3 sec
