A) \[C{{H}_{3}}C{{H}_{2}}OH\]
B) \[C{{H}_{3}}CHOH\cdot C{{H}_{3}}\]
C) \[{{(C{{H}_{3}})}_{3}}C\cdot CHO\]
D) \[HO-C{{H}_{2}}\cdot C{{H}_{2}}-OH\]
Correct Answer: B
Solution :
Alcohols shows weak acidic character, when treated with active metals like K or Na and evolve hydrogen. On oxidation, secondary alcohol yield ketone. Hence, the compound must be \[C{{H}_{3}}CHOH\cdot C{{H}_{3}}.\]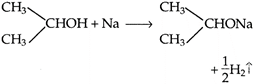
You need to login to perform this action.
You will be redirected in
3 sec
