A) benzaldehyde
B) toluene
C) benzoic acid
D) ethyl benzene
Correct Answer: C
Solution :
Toluene, gives different oxidation products, with different oxidizing agents. With \[Cr{{O}_{2}}C{{l}_{2}},\] it gives benzaldehyde, while with \[KMn{{O}_{4}}\text{/}{{H}^{+}}\] or \[{{K}_{2}}C{{r}_{2}}{{O}_{7}}\text{/}{{H}^{+}},\]it gives benzoic acid.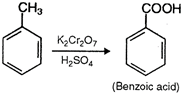
You need to login to perform this action.
You will be redirected in
3 sec
