A) 30%
B) 32%
C) 34%
D) 28%
Correct Answer: B
Solution :
No. of effective atom in a unit cell in b.c.c. = 2 Let side length of the cube\[=a\] from b.c.c. structure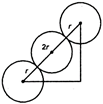 \[4r=\sqrt{3a}\] or, \[a=\frac{4r}{\sqrt{3}}\] Packing fraction\[=\frac{2\times \frac{4}{3}\pi {{(r)}^{3}}}{{{\left( \frac{4r}{\sqrt{3}} \right)}^{3}}}\times 100\] \[=\frac{\sqrt{3\pi }}{8}\times 100=68%\] Percentage of free space = 32%.
\[4r=\sqrt{3a}\] or, \[a=\frac{4r}{\sqrt{3}}\] Packing fraction\[=\frac{2\times \frac{4}{3}\pi {{(r)}^{3}}}{{{\left( \frac{4r}{\sqrt{3}} \right)}^{3}}}\times 100\] \[=\frac{\sqrt{3\pi }}{8}\times 100=68%\] Percentage of free space = 32%.
You need to login to perform this action.
You will be redirected in
3 sec
