A) \[{{[CoF]}^{3-}}\]
B) \[{{[CO{{(N{{H}_{3}})}_{6}}]}^{3+}}\]
C) \[{{[Fe{{(CN)}_{6}}]}^{3-}}\]
D) \[{{[Cr{{(N{{H}_{3}})}_{6}}]}^{3+}}\]
Correct Answer: A
Solution :
(i) Electronic configuration of\[C{{o}^{3+}}\]ion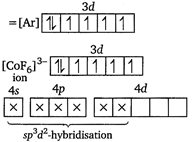 \[{{F}^{-}}\]is a weak ligand. It cannot pair up electrons within d-subshell and forms outer orbital octahedral complex. (ii)\[{{[Co{{(N{{H}_{3}})}_{6}}]}^{3+}}\]ion
\[{{F}^{-}}\]is a weak ligand. It cannot pair up electrons within d-subshell and forms outer orbital octahedral complex. (ii)\[{{[Co{{(N{{H}_{3}})}_{6}}]}^{3+}}\]ion 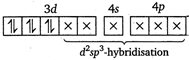 (iii)\[{{[Fe{{(CN)}_{6}}]}^{3-}}\]ion
(iii)\[{{[Fe{{(CN)}_{6}}]}^{3-}}\]ion 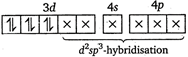 (iv)\[{{[Cr{{(N{{H}_{3}})}_{6}}]}^{3+}}\]ion
(iv)\[{{[Cr{{(N{{H}_{3}})}_{6}}]}^{3+}}\]ion 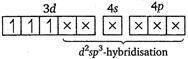 \[N{{H}_{3}}\]and\[C{{N}^{-}}\]are strong ligands. So, they form inner orbital complex.
\[N{{H}_{3}}\]and\[C{{N}^{-}}\]are strong ligands. So, they form inner orbital complex.
You need to login to perform this action.
You will be redirected in
3 sec
