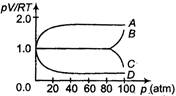
A) Curve A
B) Curve B
C) Curve C
D) Curve D
Correct Answer: B
Solution :
At lower pressure, we can assume that given gas behaves as ideal gas so \[5.6\times {{10}^{-7}}m\] constant but when pressure increase, -the decrease in volume will not take place in same proportion so\[{{10}^{-6}}{{m}^{2}},\]will increases.You need to login to perform this action.
You will be redirected in
3 sec
