A) the electronegativity of F is greater than that of 0
B) \[{{H}_{2}}O\]involves hydrogen bonding whereas \[Be{{F}_{2}}\]is a discrete molecule
C) \[{{H}_{2}}O\]is linear and\[Be{{F}_{2}}\]is angular
D) \[{{H}_{2}}O\]is angular and\[Be{{F}_{2}}\]is linear
Correct Answer: D
Solution :
The structure of\[{{H}_{2}}O\]is angular V-shape and has\[s{{p}^{3}}-\]hybridisation and bond angle is 105°. Its dipole moment value is positive or more than zero.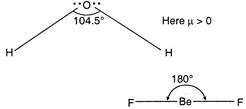 but in\[Be{{F}_{2}},\]structure is linear due to\[sp-\]hybridisation\[(\mu =0)\] Thus, due to\[\mu >0,{{H}_{2}}O\]is dipolar and due to \[\mu =0,\text{ }Be{{F}_{2}}\]is non-polar.
but in\[Be{{F}_{2}},\]structure is linear due to\[sp-\]hybridisation\[(\mu =0)\] Thus, due to\[\mu >0,{{H}_{2}}O\]is dipolar and due to \[\mu =0,\text{ }Be{{F}_{2}}\]is non-polar.
You need to login to perform this action.
You will be redirected in
3 sec
