A) \[C{{H}_{3}}OH+{{(C{{H}_{3}})}_{3}}Cl\]
B) \[C{{H}_{3}}I+{{(C{{H}_{3}})}_{3}}COH\]
C) \[C{{H}_{3}}I+{{(C{{H}_{3}})}_{3}}Cl\]
D) None of the above
Correct Answer: B
Solution :
Here, ether is the solvent. Being less polar, it favours\[{{S}_{N}}2\]reaction and the nucleophile F attacks the\[1{}^\circ \]carbon of\[C{{H}_{3}}\].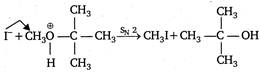
You need to login to perform this action.
You will be redirected in
3 sec
