A) \[C{{H}_{3}}COOH\]
B) \[C{{H}_{3}}COCl\]
C) \[{{(C{{H}_{3}}CO)}_{2}}O\]
D) All the above three
Correct Answer: D
Solution :
When alcohols react with acids or acid derivatives, esters are formed. Thus, \[C{{H}_{3}}OH+C{{H}_{3}}COOH\xrightarrow[{}]{{}}\underset{methyl\text{ }acetate}{\mathop{C{{H}_{3}}COOC{{H}_{3}}}}\,+{{H}_{2}}O\]; \[C{{H}_{3}}OH+C{{H}_{3}}COCl\xrightarrow[{}]{{}}\underset{methyl\text{ }acetate}{\mathop{C{{H}_{3}}COOC{{H}_{3}}}}\,+HCl\]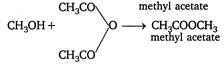 \[+C{{H}_{3}}COOH\]
\[+C{{H}_{3}}COOH\]
You need to login to perform this action.
You will be redirected in
3 sec
