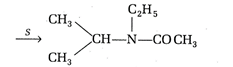
 Which of the following are P and S in the above series of reactions?
Which of the following are P and S in the above series of reactions?
A) \[{{C}_{2}}{{H}_{5}}MgX\]and \[{{({{C}_{2}}{{H}_{5}})}_{2}}NC{{H}_{3}}\]
B) \[C{{H}_{3}}MgX\] and N-methyl -2 ? butanamine
C) \[C{{H}_{3}}MgX\] and N-ethyl ? 2 butanamine
D) \[{{C}_{2}}{{H}_{5}}MgX\]and \[{{(C{{H}_{3}})}_{2}}N{{C}_{2}}{{H}_{5}}\]
Correct Answer: C
Solution :
\[C{{O}_{2}}+\underset{(P)}{\mathop{C{{H}_{3}}MgX}}\,\xrightarrow{{{H}_{2}}O}\underset{(Q)}{\mathop{C{{H}_{3}}COOH}}\,\xrightarrow[-{{H}_{2}}O]{{{P}_{2}}{{O}_{5}}}\]\[\begin{align} & NH-{{C}_{2}}{{H}_{5}} \\ & | \\ & \underset{(R)}{\mathop{{{(C{{H}_{3}}CO)}_{2}}}}\,O\xrightarrow[(S)]{C{{H}_{3}}-CH-{{C}_{2}}{{H}_{5}}} \\ \end{align}\]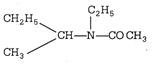
You need to login to perform this action.
You will be redirected in
3 sec
