A) -5J
B) -10J
C) -15J
D) -20J
Correct Answer: A
Solution :
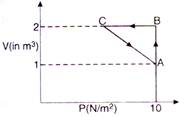
Work done in Process\[A\to B\] \[{{W}_{AB}}=P\Delta V\] \[=10(2-1)=10J\]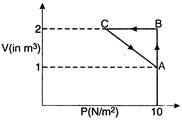 Process\[B\to C\]is at constant volume so work done is zero. The process is cyclic, so change in internal energy \[\Delta U=0\] From 1st law of thermodynamics \[\Delta Q=\Delta U+W\] \[5=\Delta ({{W}_{AB}}+{{W}_{BC}}+{{W}_{CA}})\] \[5=10+0+{{W}_{CA}}\] \[\therefore \] \[{{W}_{CA}}=-5J\]
Process\[B\to C\]is at constant volume so work done is zero. The process is cyclic, so change in internal energy \[\Delta U=0\] From 1st law of thermodynamics \[\Delta Q=\Delta U+W\] \[5=\Delta ({{W}_{AB}}+{{W}_{BC}}+{{W}_{CA}})\] \[5=10+0+{{W}_{CA}}\] \[\therefore \] \[{{W}_{CA}}=-5J\]
You need to login to perform this action.
You will be redirected in
3 sec
