A) steric hindrance
B) inductive effect
C) instability
D) insolubility
Correct Answer: A
Solution :
In\[{{S}_{N}}2\]reaction, nucleophile and alkyi halide react in one step.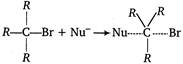 Here, it is evident that tertiary carbon is under steric hindrance, thus reaction does not take place until\[(CBr)\]bond breaks\[R-\underset{\begin{smallmatrix} | \\ R \end{smallmatrix}}{\overset{\begin{smallmatrix} R \\ | \end{smallmatrix}}{\mathop{C}}}\,-Br\xrightarrow{Slow}R-\underset{\begin{smallmatrix} | \\ R \end{smallmatrix}}{\overset{\begin{smallmatrix} R \\ | \end{smallmatrix}}{\mathop{{{C}^{\oplus }}}}}\,-B{{r}^{-}}\]which is then\[{{S}_{N}}1\]reaction.
Here, it is evident that tertiary carbon is under steric hindrance, thus reaction does not take place until\[(CBr)\]bond breaks\[R-\underset{\begin{smallmatrix} | \\ R \end{smallmatrix}}{\overset{\begin{smallmatrix} R \\ | \end{smallmatrix}}{\mathop{C}}}\,-Br\xrightarrow{Slow}R-\underset{\begin{smallmatrix} | \\ R \end{smallmatrix}}{\overset{\begin{smallmatrix} R \\ | \end{smallmatrix}}{\mathop{{{C}^{\oplus }}}}}\,-B{{r}^{-}}\]which is then\[{{S}_{N}}1\]reaction.
You need to login to perform this action.
You will be redirected in
3 sec
