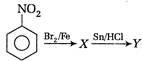
 The products X and Y respectively are
The products X and Y respectively are
A) a-bromo nitrobenzene and o-bromoaniline
B) p-bromo nitrobenzene and p-bromoaniline
C) m-bromo nitrobenzene and m-bromoaniline
D) o, p-dibromonitrobenzene and o, p-dibromoaniline
Correct Answer: C
Solution :
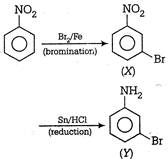 Nitro group is a deactivating and m-directing group hence it direct the incoming\[B{{r}^{-}}\]ion to the\[m-\]position, thus leading to the formation of\[m-\]bromonitrobenzene. By reduction with \[Sn/HCl,\]the nitro group reduces to\[-N{{H}_{2}}\] i.e. the final product is m-bromoaniline.
Nitro group is a deactivating and m-directing group hence it direct the incoming\[B{{r}^{-}}\]ion to the\[m-\]position, thus leading to the formation of\[m-\]bromonitrobenzene. By reduction with \[Sn/HCl,\]the nitro group reduces to\[-N{{H}_{2}}\] i.e. the final product is m-bromoaniline.
You need to login to perform this action.
You will be redirected in
3 sec
