| In a set of the given reactions, acetic acid yielded a product C, |
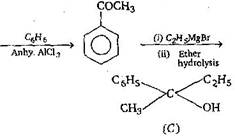
| \[C{{H}_{3}}COOH+PC{{l}_{5}}\to A\xrightarrow[Anh.\,AlC{{l}_{3}}]{{{C}_{6}}{{H}_{6}}}\]\[B\underset{Ether}{\mathop{\xrightarrow{{{C}_{2}}{{H}_{5}}MgBr}}}\,C\] product C would be : [AIPMT 2003] |
A) \[C{{H}_{3}}CH(OH){{C}_{6}}{{H}_{5}}\]
B) \[C{{H}_{3}}-\overset{\begin{smallmatrix} {{C}_{2}}{{H}_{5}} \\ | \end{smallmatrix}}{\mathop{C(OH){{C}_{6}}{{H}_{5}}}}\,\]
C) \[C{{H}_{3}}CH(OH){{C}_{2}}{{H}_{5}}\]
D) \[C{{H}_{3}}CO{{C}_{6}}{{H}_{5}}\]
Correct Answer: B
Solution :
| [b] \[C{{H}_{3}}COOH+PC{{l}_{5}}\xrightarrow[{}]{{}}C{{H}_{3}}\underset{(A)}{\mathop{COCl}}\,\] |
 |
You need to login to perform this action.
You will be redirected in
3 sec
