A) \[-\text{ }0.75,\text{ }0.6\]
B) \[-\text{ }0.75,\text{ }1.0\]
C) \[-\text{ }0.75,\text{ }1.25\]
D) \[-\text{ }3,\text{ }1.25\]
Correct Answer: C
Solution :
| Bond order between \[P-O\] |
| \[=\frac{\text{no}\text{. of bonds in all possible direction}}{\text{Total no}\text{. of resonating structure}}\] |
| \[=\frac{5}{4}=1.25\] |
| Resonating structures are : |
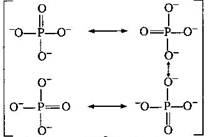 |
| Total charge on \[PO_{4}^{3-}\] ion is \[-3\]. |
| So, the average formal charge on each \['O'\] atom is \[=-\frac{3}{4}=-0.75\] |
You need to login to perform this action.
You will be redirected in
3 sec
