A) \[Si{{F}_{4}}\] and \[S{{F}_{4}}\]
B) \[IO_{3}^{-}\] and \[Xe{{O}_{3}}\]
C) \[B{{H}_{4}}\] and \[NH_{4}^{+}\]
D) \[PF_{6}^{-}\] and \[SF_{6}^{{}}\]
Correct Answer: A
Solution :
| \[Si{{F}_{4}}\] and \[S{{F}_{4}}\] are not is structural because \[Si{{F}_{4}}\] is tetrahedral due to \[\text{s}{{\text{p}}^{3}}\]hybridisation of Si. |
| \[_{14}Si=1{{s}^{2}},2{{s}^{2}},2{{p}^{6}},3{{s}^{2}}3{{p}^{2}}\] (In ground state) |
| \[_{14}Si=1{{s}^{2}},2{{s}^{2}},2{{p}^{6}},3{{s}^{2}}3{{p}^{2}}\] (In excited state) |
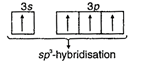 |
| \[_{14}Si=1{{s}^{2}},2{{s}^{2}},2{{p}^{6}},3{{s}^{1}}3{{p}^{3}}\] (In excited state) |
| Hence, four equivalent\[s{{p}^{3}}\]-hybrid orbitals are obtained and they are overlapped by four p-orbitals of four fluorine atoms on their axes. |
| Thus it shows following structure : |
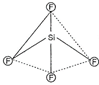 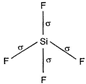 |
| While \[S{{F}_{4}}\] is not tetrahedral but it is distorted tetrahedral because in it S is \[s{{p}^{3}}\] d-hybrid. |
| \[_{16}S=1{{s}^{2}},2{{s}^{2}}2{{p}^{6}},3{{s}^{2}}3p_{x}^{2}3p_{y}^{1}3p_{z}^{1}\] |
| (In ground state) |
| \[=1{{s}^{2}},2{{s}^{2}}2{{p}^{6}},3{{s}^{2}}3p_{x}^{1}3p_{y}^{1}3p_{z}^{1,}3d_{xy}^{1}\] |
| \[s{{p}^{3}}\] d-hybridisation |
| (In first excitation state) |
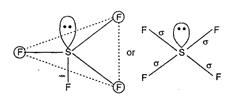 |
| Hence, five \[s{{p}^{3}}\] d-hybrid orbitals are obtained. |
| One orbital is already paired and rest four are overlapped with four p-orbitals of four fluorine atoms on their axis in trigonal bipyramidal form. This structure is distorted from trigonal bi-pyramidal to tetrahedral due to involvement of repulsion between lone pair and bond pair. |
You need to login to perform this action.
You will be redirected in
3 sec
