A) \[CO_{3}^{2-},SO_{3}^{2-}\]
B) \[ClO_{3}^{-},\,CO_{3}^{2-}\]
C) \[SO_{3}^{2-},NO_{3}^{-}\]
D) \[ClO_{3}^{-},SO_{3}^{2-}\]
Correct Answer: D
Solution :
| \[CIO_{3}^{-},=SO_{3}^{2-}\] |
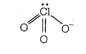 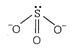 |
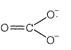 |
| Number of electrons |
| \[CO_{3}^{2-}=6+2+24=32\] |
| \[SO_{3}^{2-}=16+2+24=42\] |
| \[CIO_{3}^{-}=17+24+1=42\] |
| \[CO_{3}^{2-}=6+24+2=32\] |
| \[NO_{3}^{2-}=7+2+24=33\] |
| Hence, \[CIO_{3}^{-}\] and \[SO_{3}^{2-}\] are isoelectronic and are pyramidal in shape. |
You need to login to perform this action.
You will be redirected in
3 sec
