A) \[{{H}_{2}}O\]
B) \[B{{F}_{3}}\]
C) \[NH_{2}^{-}\]
D) \[PC{{l}_{3}}\]
Correct Answer: D
Solution :
| [a] \[{{H}_{2}}O\Rightarrow \] |
| |
[b] 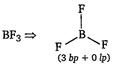 |
[c] 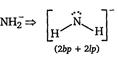 |
[d] 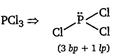 |
| Thus, in PCl3, the central atom P ha:, three bond pairs and one lone pair. |
You need to login to perform this action.
You will be redirected in
3 sec
