| Which of the following complex compounds will exhibit highest paramagnetic behaviour? [AIPMT (M) 2011] |
| \[(At.\,no.:Ti=22,Cr=24,Co=27,Zn=30)\] |
A) \[{{[Ti{{(N{{H}_{3}})}_{6}}]}^{3+}}\]
B) \[{{[Cr{{(N{{H}_{3}})}_{6}}]}^{3+}}\]
C) \[{{[Co{{(N{{H}_{3}})}_{6}}]}^{3+}}\]
D) \[{{[Zn{{(N{{H}_{3}})}_{6}}]}^{2+}}\]
Correct Answer: C
Solution :
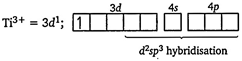
| [c] [a] Electronic configuration of \[\text{T}{{\text{i}}^{\text{3+}}}\] in |
| \[{{[Ti{{(N{{H}_{3}})}_{6}}]}^{3+}}\] |
 |
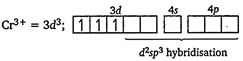
| [b] Electronic configuration of \[\text{C}{{\text{r}}^{\text{3+}}}\]in\[{{\text{ }\!\![\!\!\text{ Cr(N}{{\text{H}}_{\text{3}}}{{\text{)}}_{\text{6}}}\text{ }\!\!]\!\!\text{ }}^{\text{3+}}}\] |
 |
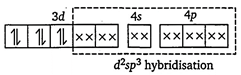
| [c] Electronic configuration of \[\text{C}{{\text{O}}^{\text{3+}}}\]in\[{{\text{ }\!\![\!\!\text{ Co(N}{{\text{H}}_{\text{3}}}{{\text{)}}_{\text{6}}}\text{ }\!\!]\!\!\text{ }}^{\text{3+}}}\text{:}\] |
| |
| In the presence of strong field ligand \[N{{H}_{3}},\]pairing of electrons takes place and thus, octahedral complex,\[{{\text{ }\!\![\!\!\text{ Co(N}{{\text{H}}_{\text{3}}}{{\text{)}}_{\text{6}}}\text{ }\!\!]\!\!\text{ }}^{\text{3+}}}\] is diamagnetic. \[{{\text{ }\!\![\!\!\text{ Co(N}{{\text{H}}_{\text{3}}}{{\text{)}}_{\text{6}}}\text{ }\!\!]\!\!\text{ }}^{\text{3+}}}\] |
| \[\left[ \begin{align} & \text{inner}\,\text{orbital}\,\text{or} \\ & \text{low}\,\text{spin}\,\text{complex} \\ \end{align} \right]\text{(6N}{{\text{H}}_{\text{3}}}\,\text{molecules)}\] |
 |
| [d] Electronic configuration of\[Z{{n}^{2+}}\] in |
| \[{{[Zn{{(N{{H}_{3}})}_{6}}]}^{2+}}:\] |
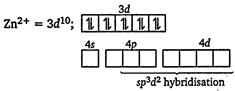 |
| \[\therefore \]\[{{[Zn{{(N{{H}_{3}})}_{6}}]}^{2+}}\]is an outer orbital complex and is diamagnetic. |
You need to login to perform this action.
You will be redirected in
3 sec
