A) \[[Co{{(en)}_{2}}C{{l}_{2}}]Cl\]
B) \[[Co(en)(N{{H}_{3}})C{{l}_{2}}]Cl\]
C) \[[Co{{(PP{{H}_{3}})}_{2}}(N{{H}_{3}})C{{l}_{2}}]Cl\]
D) \[[Co{{(en)}_{3}}]C{{l}_{3}}\]
Correct Answer: B
Solution :
| [b] Complex \[[Co\,(en)\,{{(N{{H}_{3}})}_{2}}C{{l}_{2}}]\text{ }Cl\] will have four different isomers. |
| (i) Geometrical isomers. |
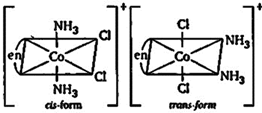 |
| (ii) Optical isomers |
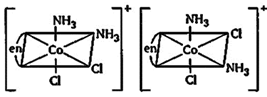 |
You need to login to perform this action.
You will be redirected in
3 sec
