| Which one of the following is an inner orbital complex as well as diamagnetic in behaviour? [AIPMT (S) 2005] |
| (Atomic no. : Zn = 30, Cr=24, Co=27, Ni = 28) |
A) \[{{[Zn\,{{(N{{H}_{3}})}_{6}}]}^{2+}}\]
B) \[{{[Cr{{(N{{H}_{3}})}_{6}}]}^{3+}}\]
C) \[{{[Co{{(N{{H}_{3}})}_{6}}]}^{3+}}\]
D) \[{{[Ni\,{{(N{{H}_{3}})}_{6}}]}^{2+}}\]
Correct Answer: B
Solution :
| [b] In [Co(NH3)6]3+, oxidation state of Co = + 3 and its co-ordination number is six. |
| So \[_{27}Co=1{{s}^{2}},2{{s}^{2}},2{{p}^{6}},3{{s}^{2}}3{{p}^{6}},3{{d}^{7}},4{{s}^{2}}\] |
| \[C{{o}^{3+}}=1{{s}^{2}},2{{s}^{2}}2{{p}^{6}},3{{s}^{2}}3{{p}^{6}}3{{d}^{6}}\] |
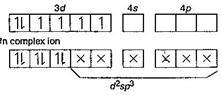 |
| In [Co(NH3)6]3+ shows inner orbital complex as well as diamagnetic in behaviour (due to absence of upaired electron). |
| [Zn(NH3)6]3+ \[\to \] sp3d2 hybridisation (outer) and diamagnetic. |
| \[{{[Cr{{(N{{H}_{3}})}_{6}}]}^{2+}}\to \,{{d}^{2}}s{{p}^{3}}\] hybridisation (inner) and paramagnetic. |
You need to login to perform this action.
You will be redirected in
3 sec
