A) \[ds{{p}^{2}}\]
B) \[s{{p}^{3}}\]
C) \[{{d}^{2}}s{{p}^{2}}\]
D) \[{{d}^{2}}s{{p}^{3}}\]
Correct Answer: A
Solution :
| [a] \[{{[Ni(CN)4]}^{2-}}\] |
| Let oxidation state of Ni in \[{{[Ni(CN)4]}^{2-}}\]is x. |
| \[\therefore \] \[x-4=2\] |
| or x = 2 |
| Now, \[N{{i}^{2+}}=[Ar],3{{d}^{8}},4{{s}^{0}}\] |
| |
| \[\because \]\[C{{N}^{-}}\] is a strong field ligand. Hence, all unpaired electrons are paired up. |
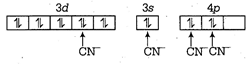 |
| \[\therefore \] Hybridisation of \[{{[Ni{{(CN)}_{2}}]}^{2-}}\]is \[ds{{p}^{2}}\] |
You need to login to perform this action.
You will be redirected in
3 sec
