A) \[\frac{2}{3}\]
B) \[\frac{3}{4}\]
C) 2
D) \[\frac{1}{2}\]
Correct Answer: B
Solution :
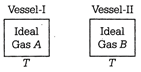 |
| \[{{\rho }_{A}}=1.5{{\rho }_{B}}\] \[{{\rho }_{B}}\] |
| \[{{\rho }_{A}}=2{{\rho }_{B}}\] \[{{p}_{B}}\] |
| According to ideal gas equation, we have |
| Pressure, \[p=\frac{\rho RT}{M},\] where M is molecular weight of ideal gas. |
| Such that, \[\frac{p}{\rho }=\frac{RT}{M}\Rightarrow M=\frac{\rho RT}{p}\] |
| where, R and T are constant. |
| So, \[M\propto \frac{\rho }{p}\] |
| \[\Rightarrow \] \[\frac{{{M}_{A}}}{{{M}_{B}}}=\frac{{{\rho }_{A}}}{{{\rho }_{B}}}\times \frac{{{p}_{B}}}{{{p}_{A}}}=1.5\times \frac{1}{2}=0.75=\frac{3}{4}\] |
You need to login to perform this action.
You will be redirected in
3 sec
