A)
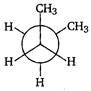
B)
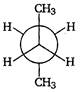
C)
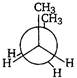
D)
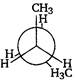
Correct Answer: B
Solution :
| Key Idea The conformation in which the heavier groups are present at maximum possible distances, so that the forces of repulsion get weak, is more stable. |
| Among the given conformations of n-butane, the conformation shown in option i.e., anti-conformation is most stable as in it the bulkier group (i.e., \[C{{H}_{3}}\] group) are present at maximum possible distance. |
You need to login to perform this action.
You will be redirected in
3 sec
