| In which of the following pairs are both the ions coloured in aqueous solution? [AIPMT (S) 2006] |
| (At. no.: Sc = 21, Ti = 22, Ni = 28, Cu = 29, Co = 27) |
A) Ni2+, Ti3+
B) Sc3+, Ti3+
C) Sc3 , Co2+
D) Ni2+, Cu+
Correct Answer: A
Solution :
| [a] \[_{28}Ni=1{{s}^{2}},2{{s}^{2}}\,2{{p}^{6}},3{{s}^{2}}3{{p}^{6}}3{{d}^{8}},4{{s}^{2}}\] |
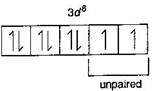 |
| \[N{{i}^{2+}}=1{{s}^{2}},2{{s}^{2}}2{{p}^{6}},3{{s}^{2}}3{{p}^{6}}3{{d}^{{}}}\] |
| \[_{22}Ti=1{{s}^{2}},2{{s}^{2}}2{{p}^{6}},3{{s}^{2}}3{{p}^{6}}3{{d}^{2}},4{{s}^{2}}\] |
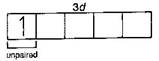 |
| \[T{{i}^{3+}}=1{{s}^{2}},2{{s}^{2}}2{{p}^{6}},3{{s}^{2}}3{{p}^{6}}3{{d}^{1}}\] |
| \[_{21}Sc=1{{s}^{2}},2{{s}^{2}}2{{p}^{6}},3{{s}^{2}}3{{p}^{6}}3{{d}^{1}},4{{s}^{2}}\] |
| \[S{{c}^{3+}}=1{{s}^{2}},2{{s}^{2}}2{{p}^{6}},3{{s}^{2}}3{{p}^{6}}\] |
| (unpaired electron in d-orbital is not possible) |
| \[_{29}Cu=1{{s}^{2}},2{{s}^{2}}\,2{{p}^{6}},3{{s}^{2}}\,3{{p}^{6}}\,3{{d}^{10}},4{{s}^{1}}\] |
| \[C{{u}^{+}}=1{{s}^{2}},2{{s}^{2}}2{{p}^{6}},3{{s}^{2}}3{{p}^{6}}3{{d}^{10}}\] |
| (complete d-orbital) |
| Hence, in above ions, Ni2+ and Ti3+ ions are coloured ions in aqueous solution due to presence of unpaired electrons in d-sub-shell. |
You need to login to perform this action.
You will be redirected in
3 sec
