A) Adiabatic
B) Isobaric
C) Isochoric
D) Isothermal
Correct Answer: A
Solution :
| Given, ideal gas is compressed to half its initial volume i.e. |
| \[{{V}_{0}}=\frac{V}{2}\] |
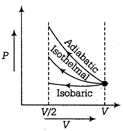 |
| The isochoric process is one in which volume is kept constant, meaning that work done by the system will be zero i.e., \[{{W}_{\text{isochoric}}}=0\] |
| As we know, work dine on the gas = Area under curve i.e. |
| \[{{W}_{\text{ediabatic}}}>{{W}_{\text{isothermal}}}>{{W}_{\text{isochoric}}}\] |
You need to login to perform this action.
You will be redirected in
3 sec
