| Thermodynamic processes are indicated in the following diagram. [NEET-2017] |
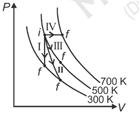 |
| Match the following |
| Column-1 Column-2 P. Process I a. Adiabatic Q. Process II b. Isobaric R. Process III c. Isochoric S. Process IV d. Isothermal |
A) \[P\to d,\,Q\to b,\,R\to a,\,S\to c\]
B) \[P\to a,\,Q\to c,\,R\to d,\,S\to b\]
C) \[P\to c,\,Q\to a,\,R\to d,\,S\to b\]
D) \[P\to c,\,Q\to d,\,R\to b,\,S\to a\]
Correct Answer: C
Solution :
| [c] Process I = Isochoric |
| II = Adiabatic |
| III = Isothermal |
| IV = Isobaric |
You need to login to perform this action.
You will be redirected in
3 sec
