| A thermodynamic system undergoes cyclic process ABCDA as shown in figure. The work done by the system in the cycle is [NEET 2014] |
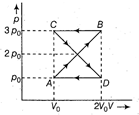 |
A) \[{{p}_{0}}{{V}_{0}}\]
B) \[2{{p}_{0}}{{V}_{0}}\]
C) \[\frac{{{p}_{0}}{{V}_{0}}}{2}\]
D) zero
Correct Answer: D
Solution :
| Work done in the cyclic process = Area bound by the closed configuration |
| = Area of closed configuration |
| \[=\left\{ 2\left[ \frac{1}{2}({{v}_{0}}/2)\times {{p}_{0}} \right]+\left\{ -2\left[ \frac{1}{2}({{v}_{0}}/2){{p}_{0}} \right] \right. \right\}\] |
| = zero. |
You need to login to perform this action.
You will be redirected in
3 sec
