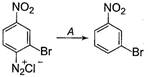
A) \[HgS{{O}_{4}}/{{H}_{2}}S{{O}_{4}}\]
B) \[C{{u}_{2}}C{{l}_{2}}\]
C) \[{{H}_{3}}P{{O}_{2}}\]and\[{{H}_{2}}O\]
D) \[{{H}^{+}}/{{H}_{2}}O\]
Correct Answer: C
Solution :
When diazonium salt is treated with\[{{\text{H}}_{\text{3}}}\text{P}{{\text{O}}_{\text{2}}}\]followed by hydrolysis, it diazonium group is replaced by ?H resulting to the formation of hydrocarbon. Thus, A must be \[{{\text{H}}_{\text{3}}}\text{P}{{\text{O}}_{\text{2}}}\text{/}\,{{\text{H}}_{\text{2}}}\text{O}\text{.}\]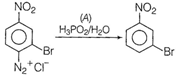
You need to login to perform this action.
You will be redirected in
3 sec
