A) \[{{N}_{3}}\]
B) \[NO_{3}^{-}\]
C) \[N{{O}_{2}}\]
D) \[C{{O}_{2}}\]
Correct Answer: B
Solution :
Species with \[s{{p}^{2}}\] hybridisation are plane triangular in shape. Among the given species \[NO_{3}^{-}\] is \[s{{p}^{2}}\] hybridised with no lone pair of electrons on central atom, N. Whereas,\[{{N}_{3}},NO_{2}^{-}\] and \[C{{O}_{2}}\] are sp hybridised with a linear shape.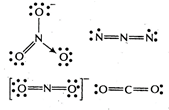
You need to login to perform this action.
You will be redirected in
3 sec
