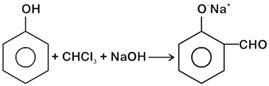
 . The electrophile involved is [NEET - 2018]
. The electrophile involved is [NEET - 2018]
A) Dichloromethyl anion \[\left( \begin{align} & \oplus \\ & \text{CHC}{{\text{l}}_{\text{2}}} \\ \end{align} \right)\]
B) Formyl cation \[\left( \begin{align} & \oplus \\ & \text{CHO} \\ \end{align} \right)\]
C) Dichloromethyl cation \[\left( \begin{align} & \oplus \\ & \text{CHC}{{\text{l}}_{2}} \\ \end{align} \right)\]
D) Dichlorocarbene \[\text{(:CC}{{\text{l}}_{\text{2}}})\]
Correct Answer: D
Solution :
It is Reimer-Tiemann reaction. The electrophile formed is: \[\text{CC}{{\text{l}}_{\text{2}}}\] (Dichlorocarbene) according to the following reaction \[\text{CHC}{{\text{l}}_{\text{3}}}\text{+O}{{\text{H}}^{\text{-}}}\overset{\begin{smallmatrix} \Theta \\ .\,\,\,\,. \end{smallmatrix}}{\mathop{\text{C}}}\,\text{C}{{\text{l}}_{3}}\text{+}{{\text{H}}_{\text{2}}}\text{O}\] \[\overset{\begin{smallmatrix} \Theta \\ .\text{ }\text{.} \end{smallmatrix}}{\mathop{\text{C}}}\,\text{C}{{\text{l}}_{\text{3}}}\xrightarrow{{}}\text{:}\underset{\text{Electrophile}}{\mathop{\text{CC}{{\text{l}}_{\text{2}}}}}\,\text{+C}{{\text{l}}^{\text{-}}}\]You need to login to perform this action.
You will be redirected in
3 sec
