A) In absence of substituents nitro group always goes to m-position.
B) In electrophilic substitution reactions amino group is meta directive.
C) Inspite of substituents nitro group always goes to only m-position.
D) In acidic (strong) medium aniline is present as anilinium ion.
Correct Answer: D
Solution :
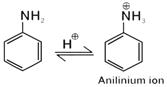 \[\text{-}\overset{\oplus }{\mathop{\text{N}}}\,{{\text{H}}_{3}}\] is m-directing, hence besides para (51%) and ortho (2%), meta product (47%) is also formed in significant yield.
\[\text{-}\overset{\oplus }{\mathop{\text{N}}}\,{{\text{H}}_{3}}\] is m-directing, hence besides para (51%) and ortho (2%), meta product (47%) is also formed in significant yield.
You need to login to perform this action.
You will be redirected in
3 sec
