A) geometrical isomerism
B) optical isomerism
C) metamerism
D) position isomerism
Correct Answer: C
Solution :
The compound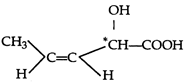 can show optical isomerism, geometrical isomerism and position isomerism. But it cannot exhibit metamerism as it has no functional group which is polyvalent.
can show optical isomerism, geometrical isomerism and position isomerism. But it cannot exhibit metamerism as it has no functional group which is polyvalent.
You need to login to perform this action.
You will be redirected in
3 sec
