A) \[NO_{3}^{-}\]
B) \[SO_{3}^{2-}\]
C) \[BO_{3}^{3-}\]
D) \[CO_{3}^{2-}\]
Correct Answer: B
Solution :
In\[SO_{3}^{2-}\], the \[S\] is \[s{{p}^{3}}\] hybridised, so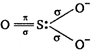 \[\underset{\begin{smallmatrix} (Sluphur\,\,atom\,\,in \\ \,\,\,\,\,\,excited\,\,state) \end{smallmatrix}}{\mathop{_{16}S=1{{s}^{2}},\,\,2{{s}^{2}}2{{p}^{6}}}}\,,\,\,\underbrace{3{{s}^{2}}3p_{x}^{1}3p_{y}^{1}3p_{z}^{1}}_{s{{p}^{3}}\,\,hybridisation}\underset{unhybridised}{\mathop{3d_{xy}^{1}}}\,\] In \[S\] the three \[p-\]orbitals forms \[\sigma \] bonds with three oxygen atoms and unhybridised \[d-\]orbital is involved in \[\pi \] bond formation \[_{16}O=1{{s}^{2}},\,\,2{{s}^{2}}2p_{x}^{2},\,\,2p_{y}^{1}2p_{z}^{1}\] In oxygen two unpaired p-orbitals are present, one is involved in \[\sigma \] bond formation while other is used in \[\pi \] bond formation. Thus in\[SO_{3}^{2-}\], \[{{p}_{\pi }}\] and \[{{d}_{\pi }}\] orbitals are involved for \[{{p}_{\pi }}-{{d}_{\pi }}\]bonding.
\[\underset{\begin{smallmatrix} (Sluphur\,\,atom\,\,in \\ \,\,\,\,\,\,excited\,\,state) \end{smallmatrix}}{\mathop{_{16}S=1{{s}^{2}},\,\,2{{s}^{2}}2{{p}^{6}}}}\,,\,\,\underbrace{3{{s}^{2}}3p_{x}^{1}3p_{y}^{1}3p_{z}^{1}}_{s{{p}^{3}}\,\,hybridisation}\underset{unhybridised}{\mathop{3d_{xy}^{1}}}\,\] In \[S\] the three \[p-\]orbitals forms \[\sigma \] bonds with three oxygen atoms and unhybridised \[d-\]orbital is involved in \[\pi \] bond formation \[_{16}O=1{{s}^{2}},\,\,2{{s}^{2}}2p_{x}^{2},\,\,2p_{y}^{1}2p_{z}^{1}\] In oxygen two unpaired p-orbitals are present, one is involved in \[\sigma \] bond formation while other is used in \[\pi \] bond formation. Thus in\[SO_{3}^{2-}\], \[{{p}_{\pi }}\] and \[{{d}_{\pi }}\] orbitals are involved for \[{{p}_{\pi }}-{{d}_{\pi }}\]bonding.
You need to login to perform this action.
You will be redirected in
3 sec
