A) \[[Cr{{(CO)}_{6}}]\]
B) \[[Fe{{(CO)}_{5}}]\]
C) \[{{[Fe{{(CN)}_{6}}]}^{4-}}\]
D) \[{{[Cr{{(N{{H}_{3}})}_{6}}]}^{3+}}\]
Correct Answer: D
Solution :
Atoms, ions or molecules having unpaired electrons are paramagnetic. In \[\mathbf{[Cr(N}{{\mathbf{H}}_{\mathbf{3}}}\mathbf{)}_{\mathbf{6}}^{\mathbf{3+}}\mathbf{]}\] is present as\[Cr(III)\]or\[C{{r}^{3+}}\] So electronic configuration is \[\underset{ground\,\,state}{\mathop{_{24}Cr=}}\,1{{s}^{2}},\,\,2{{s}^{2}}2{{p}^{6}},\,\,3{{s}^{2}}3{{p}^{6}}3{{d}^{5}},\,\,4{{s}^{1}}\] \[C{{r}^{3+}}=1{{s}^{2}},\,\,2{{s}^{2}}2{{p}^{6}},\,\,3{{s}^{2}}3{{p}^{6}}3{{d}^{3}}\]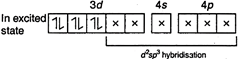 Number of unpaired electrons\[=3\] In\[\mathbf{[Cr(NO}{{\mathbf{)}}_{\mathbf{6}}}\mathbf{]}\],\[(O.N.\,\,of\,\,Cr=0)\] \[\underset{(Ground\,\,state)}{\mathop{_{24}Cr=}}\,1{{s}^{2}},\,\,2{{s}^{2}}2{{p}^{6}},\,\,3{{s}^{2}}3{{p}^{6}}3{{d}^{5}},\,\,4{{s}^{1}}\]
Number of unpaired electrons\[=3\] In\[\mathbf{[Cr(NO}{{\mathbf{)}}_{\mathbf{6}}}\mathbf{]}\],\[(O.N.\,\,of\,\,Cr=0)\] \[\underset{(Ground\,\,state)}{\mathop{_{24}Cr=}}\,1{{s}^{2}},\,\,2{{s}^{2}}2{{p}^{6}},\,\,3{{s}^{2}}3{{p}^{6}}3{{d}^{5}},\,\,4{{s}^{1}}\] 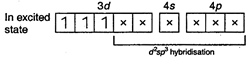 Number of unpaired electron\[=0\] In\[\mathbf{[Fe(CO}{{\mathbf{)}}_{\mathbf{5}}}\mathbf{]}\],\[(ON\,\,of\,\,Fe=0)\] \[_{26}Fe=1{{s}^{2}},\,\,2{{s}^{2}}2{{p}^{6}},\,\,3{{s}^{2}}3{{p}^{6}}3{{d}^{6}},\,\,4{{s}^{2}}\]
Number of unpaired electron\[=0\] In\[\mathbf{[Fe(CO}{{\mathbf{)}}_{\mathbf{5}}}\mathbf{]}\],\[(ON\,\,of\,\,Fe=0)\] \[_{26}Fe=1{{s}^{2}},\,\,2{{s}^{2}}2{{p}^{6}},\,\,3{{s}^{2}}3{{p}^{6}}3{{d}^{6}},\,\,4{{s}^{2}}\] 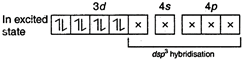 Number of unpaired electron\[=0\] In\[{{\mathbf{[Fe(CN}{{\mathbf{)}}_{\mathbf{6}}}\mathbf{]}}^{\mathbf{4-}}}\],\[(O\,\,No\,\,of\,\,Fe=+2)\] \[F{{e}^{2+}}=1{{s}^{2}},\,\,2{{s}^{2}}2{{p}^{6}},\,\,3{{s}^{2}}3{{p}^{6}}3{{d}^{6}}\]
Number of unpaired electron\[=0\] In\[{{\mathbf{[Fe(CN}{{\mathbf{)}}_{\mathbf{6}}}\mathbf{]}}^{\mathbf{4-}}}\],\[(O\,\,No\,\,of\,\,Fe=+2)\] \[F{{e}^{2+}}=1{{s}^{2}},\,\,2{{s}^{2}}2{{p}^{6}},\,\,3{{s}^{2}}3{{p}^{6}}3{{d}^{6}}\]  Number of unpaired electron\[=0\] Hence, in above complex ion paramagnetic character is in \[{{[Cr{{(N{{H}_{3}})}_{6}}]}^{3+}}\] as it contains three unpaired electrons.
Number of unpaired electron\[=0\] Hence, in above complex ion paramagnetic character is in \[{{[Cr{{(N{{H}_{3}})}_{6}}]}^{3+}}\] as it contains three unpaired electrons.
You need to login to perform this action.
You will be redirected in
3 sec
