A) \[SnC{{l}_{2}}\]and \[FeC{{l}_{3}}\]
B) \[FeC{{l}_{3}}\]and\[SnC{{l}_{4}}\]
C) \[FeC{{l}_{2}}\] and \[SnC{{l}_{4}}\]
D) \[SnC{{l}_{4}}\]and \[FeC{{l}_{3}}\]
Correct Answer: D
Solution :
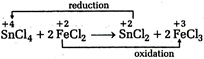 Hence,\[SnC{{l}_{4}}\]is oxidising agent while\[FeC{{l}_{2}}\]is reducing agent.
Hence,\[SnC{{l}_{4}}\]is oxidising agent while\[FeC{{l}_{2}}\]is reducing agent.
You need to login to perform this action.
You will be redirected in
3 sec
