A) phenol, p-cresol, p-nitrophenol, m-nitrophenol
B) p-cresol, phenol, 2n-nitrophenol, p-nitrophenol
C) p-cresol, m-nitrophenol, phenol, p-nitrophenol
D) m-nitrophenol, phenol, p-cresol, p-nitrophenol
Correct Answer: B
Solution :
The acidic strength of phenol increases due to the presence of electron withdrawing group (such as\[N{{O}_{2}}\]) while the acidic strength of phenol decreases due to the presence of electron releasing group (such as\[-C{{H}_{3}}\]). Hence, the order of acidic strength is: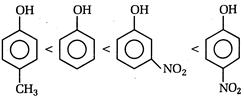
You need to login to perform this action.
You will be redirected in
3 sec
