A) \[2{{H}_{2}}S+S{{O}_{2}}\to 2{{H}_{2}}O+3S\]
B) \[Ca+{{H}_{2}}\to Ca{{H}_{2}}\]
C) \[4P+3NaOH+3{{H}_{2}}O\to P{{H}_{3}}\]\[+3Na{{H}_{2}}P{{O}_{2}}\]
D) All of the above
Correct Answer: C
Solution :
When a compound is simultaneously get reduced as well as oxidised in a reaction, then this type of reaction is called disproportionation reaction. Following reaction is an example of it.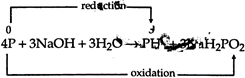
You need to login to perform this action.
You will be redirected in
3 sec
