A) \[Cl{{F}_{3}}\]
B) \[S{{O}_{3}}\]
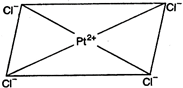
C) \[{{[Pt{{(Cl)}_{4}}]}^{2-}}\]
D) \[HgC{{l}_{2}}\]
Correct Answer: C
Solution :
\[{{[PtC{{l}_{4}}]}^{2-}}\] has square-planar geometry in it Pt is \[ds{{p}^{2}}\]-hybridized. \[_{78}Pt=[Xe]4{{f}^{14}}\,5{{d}^{8}}\,6{{s}^{2}}\] \[P{{t}^{2+}}=[Xe]\,4{{f}^{14}}\,5{{d}^{8}}\,6{{s}^{0}}\,6{{p}^{0}}\]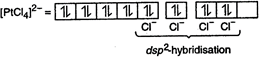
You need to login to perform this action.
You will be redirected in
3 sec
