A) \[KCl{{O}_{4}}\]
B) \[N{{H}_{4}}Cl\]
C) \[C{{H}_{3}}COONa\]
D) None of these
Correct Answer: A
Solution :
Only sails of (weak acid -+- strong base) and (strong acid -+- weak base) get hydrolyzed \[(i.e.,\]show alkalinity or acidity in water). \[KCl{{O}_{4}}\] a salt of strong acid and strong base, therefore, it does not get hydrolyzed in water. \[KCl{{O}_{4}}{{K}^{+}}+ClO_{4}^{-}\]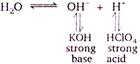
You need to login to perform this action.
You will be redirected in
3 sec
